Every cell in our body contains a copy of our genome. The human genome comprises over 20,000 genes and three billion letters of DNA. Human DNA consists of two strands, twisted into a double helix and held together by a simple pairing rule (A pairs with T, and G pairs with C).
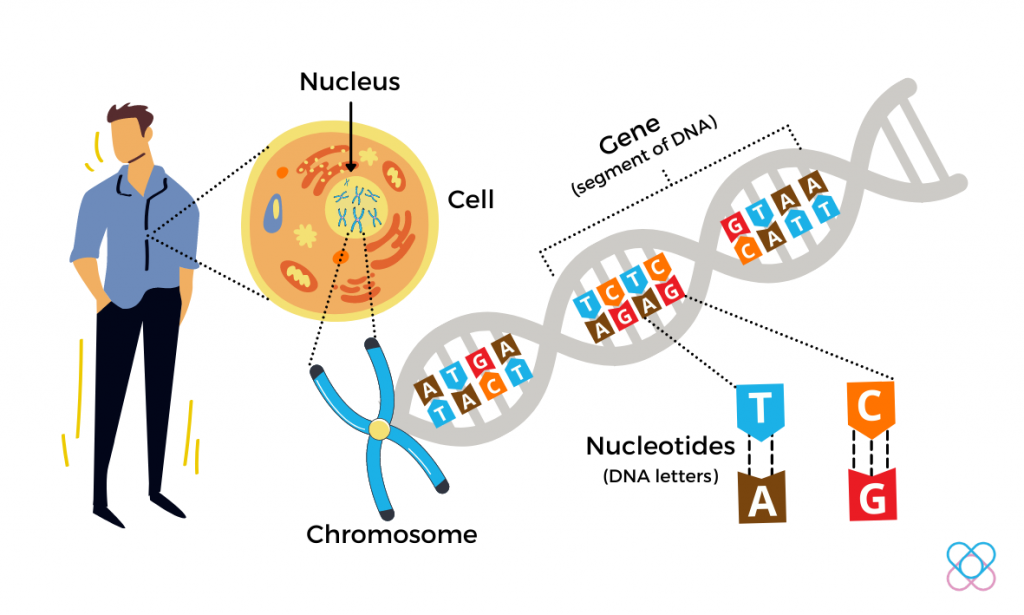
Our genes shape who we are as individuals and an intelligent species. Genes also have a profound effect on human health. Thanks to the advances in DNA sequencing technology, scientists have discovered thousands of genes that affect our disease risk. To understand how genes work, express, and eventually affect our complex biological system, scientists need ways to control and study them.
Manipulating genes in living cells is not easy. However, a recent advance in molecular sciences has developed a new method- The CRISPR-Cas9 genome editing system. This invention promises to dramatically improve our ability to edit the DNA of any species, including human2.
READ ALSO: How Genes Make Us More Susceptible To Coronaviruses
What is CRISPR-Cas9?
CRISPR-Cas9 is a powerful molecular system. It enables scientists to edit part of the genome by deleting, inserting or replacing letters of the DNA sequence.
Its application is currently recognised as the cheapest, most straightforward, versatile and precise method of genetic engineering. Therefore, it prompted a phenomenal response in the science world.
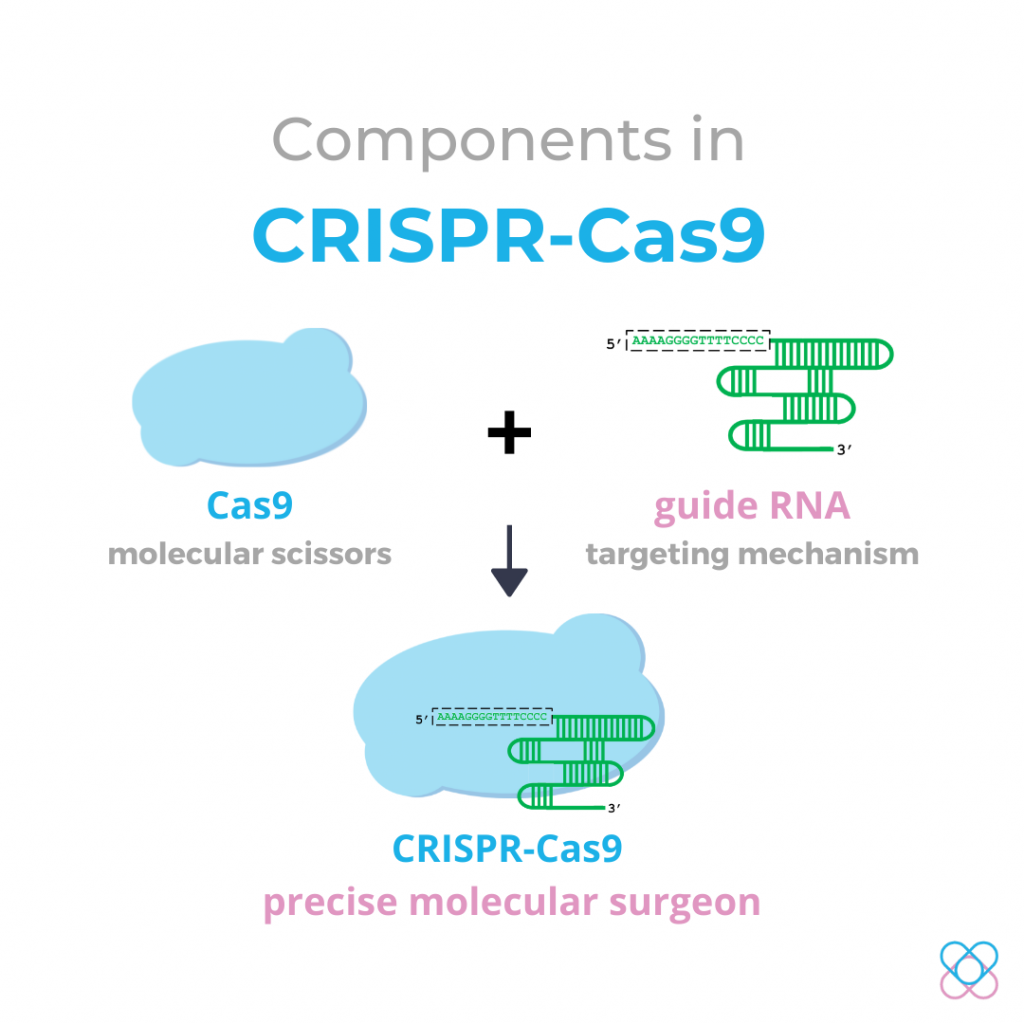
Components in the CRISPR-Cas9 system
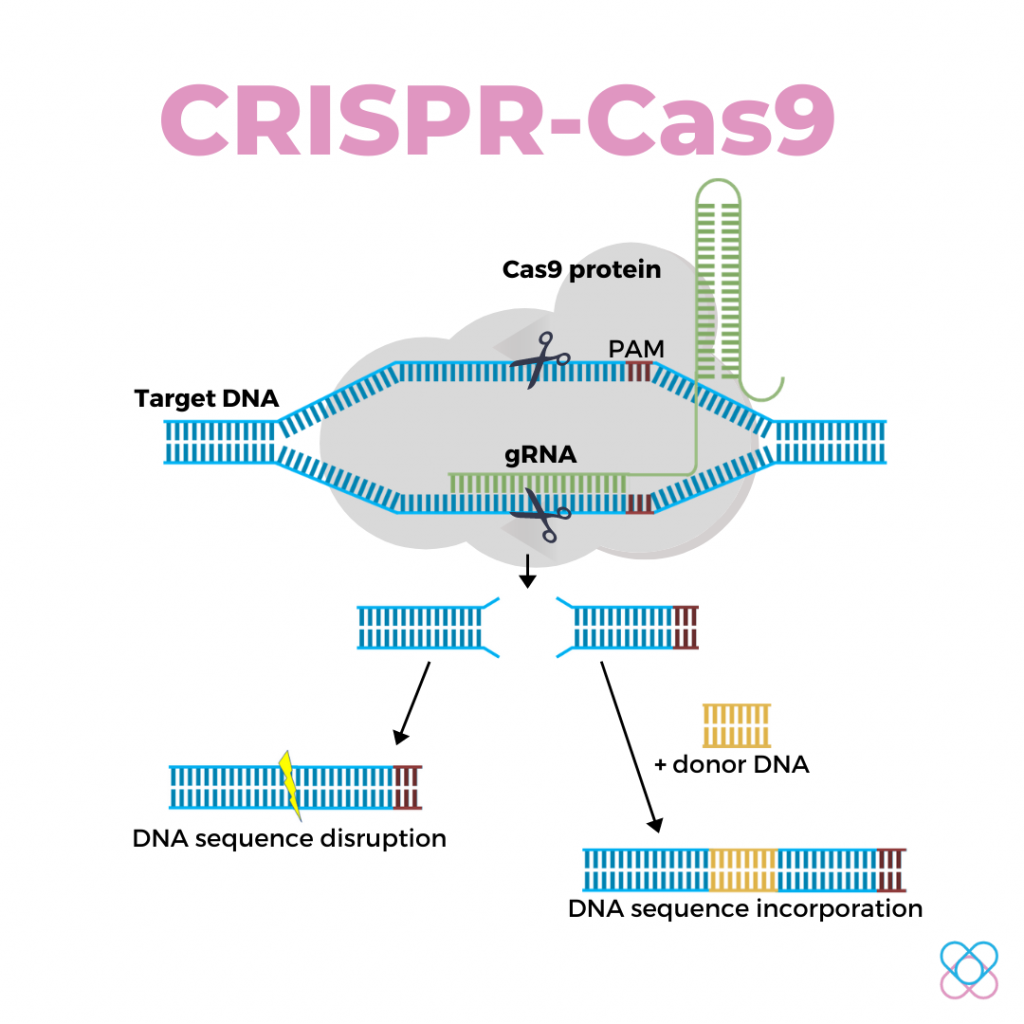
This system consists of two primary molecules that make a change into the DNA sequence2:
1. Cas9 protein
It is an enzyme that acts as a pair of molecular scissors that cut the double-stranded DNA at a precise location.
2. Guide RNA (gRNA)
It is a small piece of single-stranded sequence. It is pre-designed to match and bind to the target DNA sequence and guide the Cas9 to the right location for precise DNA cutting.
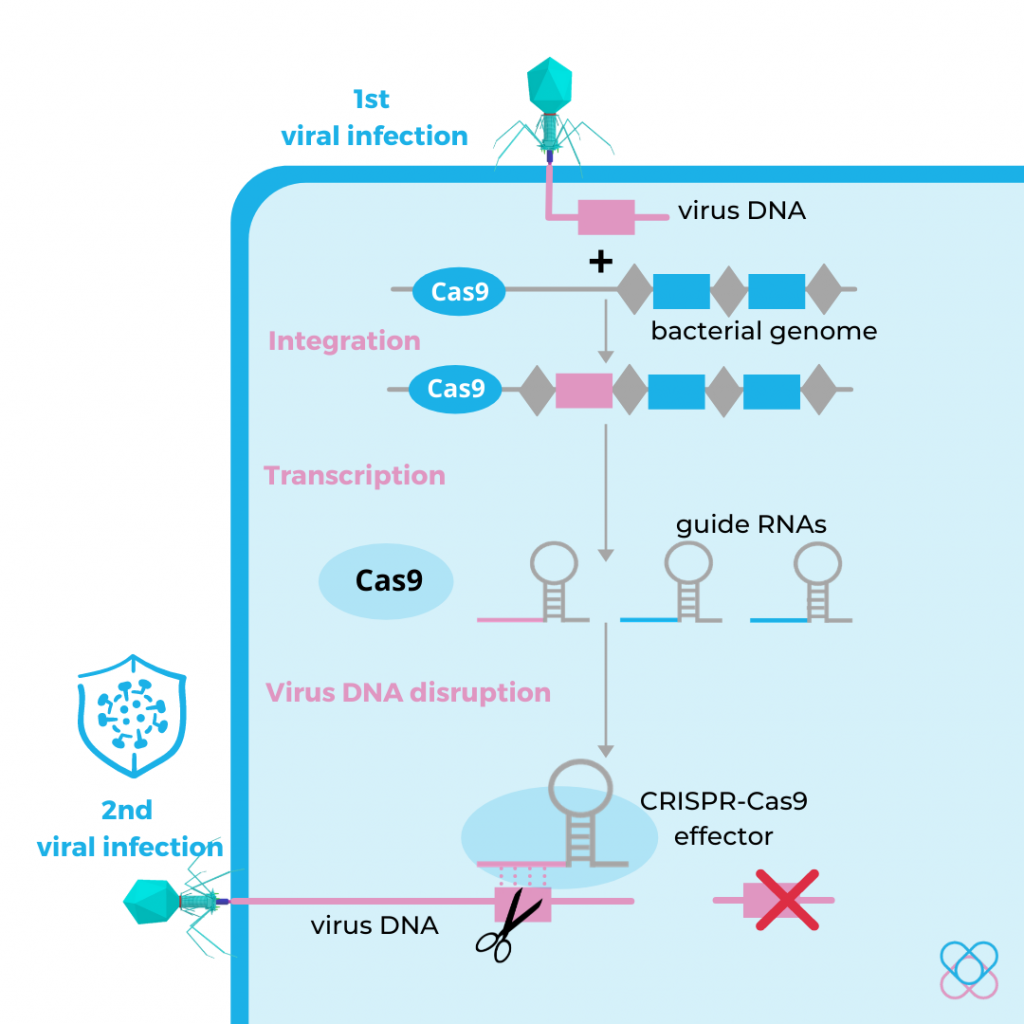
Origin of the CRISPR-Cas9 system
The development of the CRISPR-Cas9 system is based on the natural immune system used by bacteria to protect themselves from virus infection.
When a bacterium detects virus DNA, it snips out and integrates parts of the virus DNA and into its genome. This creates a memory record that can help them recognise and defend against the second virus infection by producing guide RNA (gRNA). The gRNA contains a sequence that matches that of the violating virus DNA.
This gRNA forms a complex with Cas9 protein. When the gRNA matches and binds the virus DNA, it instructs the Cas9 protein to cut the virus DNA and eventually disable the virus infection.
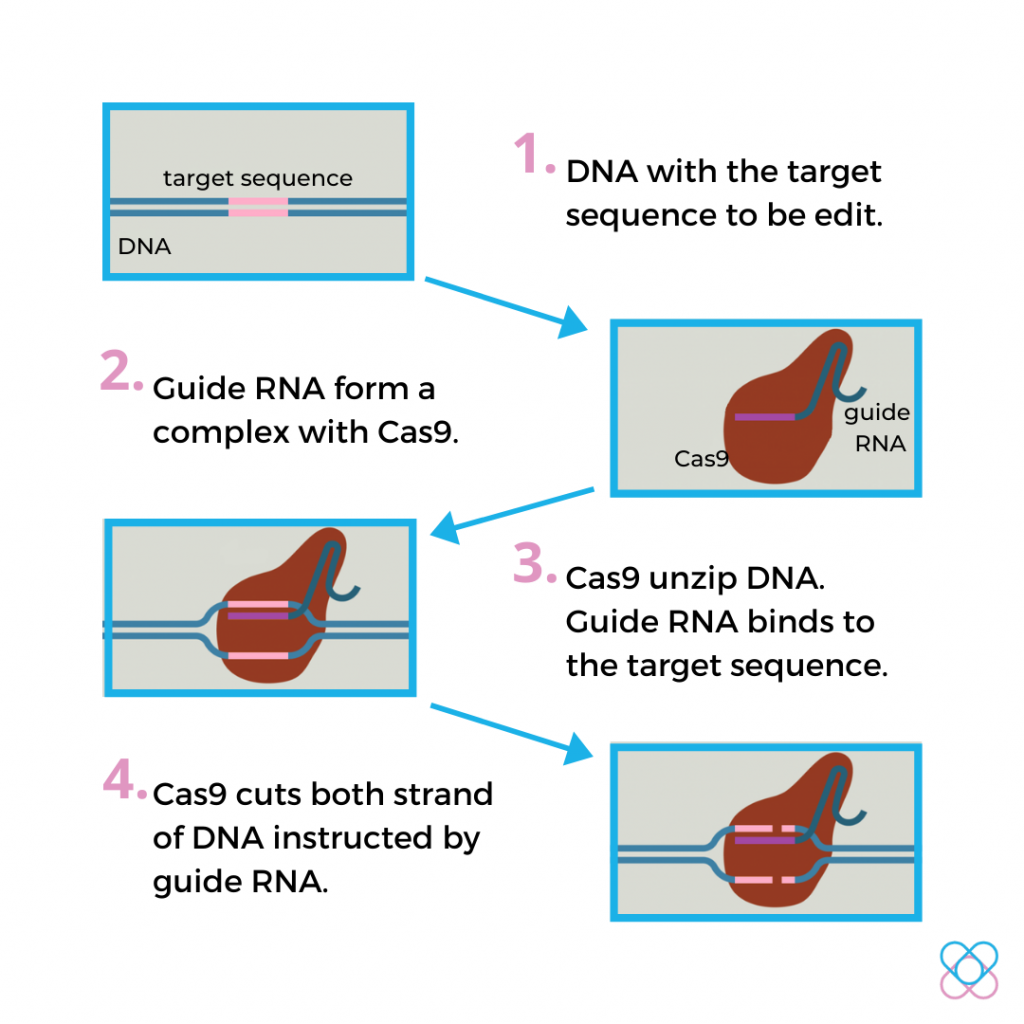
How Does It Work?
Over the past few years, scientists studied the natural system and noticed that it could be programmed to cut not just the virus DNA but any DNA sequence. It can cut DNA at a precisely chosen location by designing and modifying the gRNA.
The designed gRNA contains letters that are complementary to the target DNA, like certain functional genes.
In order words, the designed RNA will only bind specifically to the target DNA and no other regions of the genome2,3.
This system is applicable not just in a test tube but also within the nucleus of a living cell.
Once inside the nucleus, the effector CRISPR-Cas9 complex will find and lock onto a short sequence of the targeted DNA known as the PAM.
Upon locking, the Cas9 protein will unzip the double helix DNA and match it to the gRNA.
After the matching completes, gRNA guides Cas9 to cut DNA at the precise location by using its two tiny molecular scissors.
The Editing Process
The cutting process leaves a break in the double-stranded DNA, and the cell can detect and repair the break.
Upon detection, the cell will paste both broken ends together with little changes in the sequence of that position.
The cell might also integrate a new piece of DNA at the cut site.
Scientists leverage the DNA repair machinery to introduce new genes or replace defective genes with desirable ones.
Applications and Implications
CRISPR-Cas9 is notable for its ability to treat a wide range of genetic diseases. Some of which are the result of a single incorrect nucleotide in DNA.
However, the CRISPR-Cas9 system can precisely replace the single nucleotide error. This can terminate genetic diseases such as Down syndrome, Patau syndrome, Tay-Sachs disease, cystic fibrosis, and haemophilia6.
Apart from that, cancer cells that abnormally multiply while overpassing the immune response is always a big trouble.
However, an injection of CRISPR programmed immune cells can potentially target a specific cancer cell from concealing, such as lung cancer1.
Many of the applications involve the genome editing of non-reproductive cells. Nevertheless, genome programming of reproductive cells such as sperm and ovum triggers a public outcry.
This is because any genome editing in reproductive cells will be passed on from generation to generation, and it prompts ethical implications4. Therefore, genome editing in reproductive cells is illegal and prohibited in the science world.
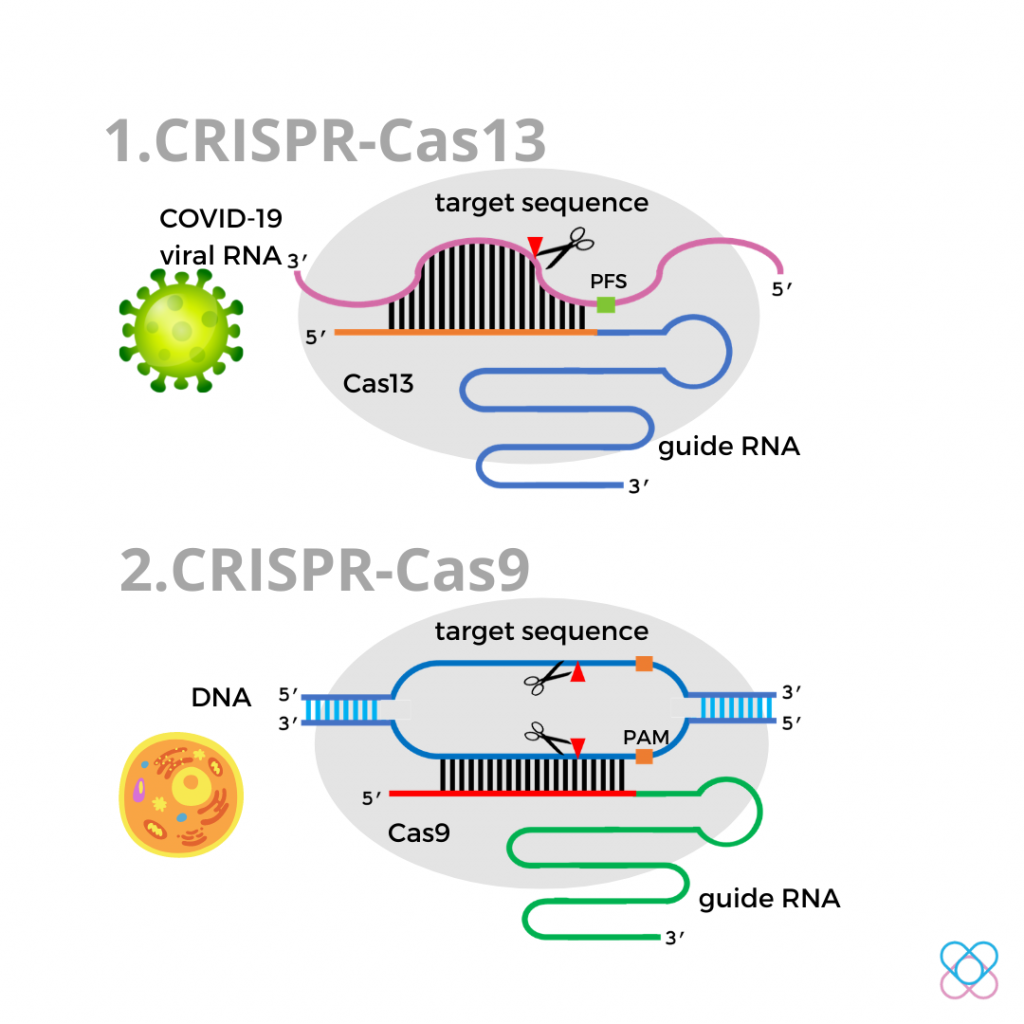
The CRISPR system and COVID-19
In parallel with the COVID-19 pandemic, a new version of the CRISPR-Cas13 system is proposed5,8. The programmed gRNA in this system targets the viral RNA genome of the SARS CoV-2 virus.
The Cas13 protein recognises the single-stranded viral RNA and cuts the RNA sequence at a specific location.
It focuses on disabling the violating viral RNA and preventing the production of infectious viral protein such as spike protein.
Thus, CRISPR-Cas13 is thought to be a potential therapy for COVID-19 aside from vaccination.
Undeniably, the potential of CRISPR in terms of scientific advances that it catalyses are beneficial for humankind. Still, it raises bioethical questions among the public.
It is a new technology, and there might have consequences and implications that remain unknown. Thus, it requires more research data and validations before practice. By the time of its extensive application, we hope to welcome the golden era that marks the end between human and diseases.
Find out your genetic predisposition in infection risk and severity when exposed to coronaviruses with Project CoviDNA today!
References
1. Castillo, A. (2016). Gene editing using CRISPR-Cas9 for the treatment of lung cancer. Colombia Médica, 47(4), 178-180.
2. Doudna, J. A., & Charpentier, E. (2014). The new frontier of genome engineering with CRISPR-Cas9. Science, 346(6213).
3.
Jiang, F., & Doudna, J. A. (2017). CRISPR–Cas9 structures and mechanisms. Annual review of biophysics, 46, 505-529.
4. Li, J. R., Walker, S., Nie, J. B., & Zhang, X. Q. (2019). Experiments that led to the first gene-edited babies: the ethical failings and the urgent need for better governance. Journal of Zhejiang University-SCIENCE B, 20(1), 32-38.
5. Lotfi, M., & Rezaei, N. (2020). CRISPR/Cas13: A potential therapeutic option of COVID-19. Biomedicine & Pharmacotherapy, 110738.
6. Pellagatti, A., Dolatshad, H., Valletta, S., & Boultwood, J. (2015). Application of CRISPR/Cas9 genome editing to the study and treatment of disease. Archives of toxicology, 89(7), 1023-1034.
7. Riordan, S. M., Heruth, D. P., Zhang, L. Q., & Ye, S. Q. (2015). Application of CRISPR/Cas9 for biomedical discoveries. Cell & bioscience, 5(1), 1-11.
8. Zhang, Y. (2020). CRISPR-Cas13 as an Antiviral Strategy for Coronavirus Disease 2019. The CRISPR Journal, 3(3), 140-142.

